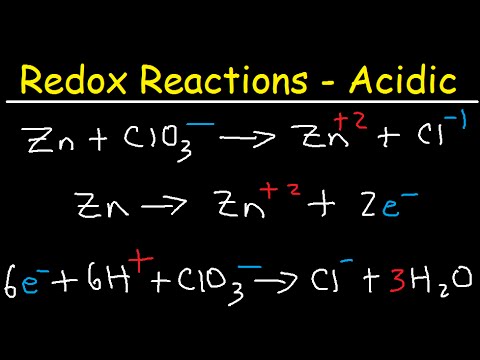 This chemistry video tutorial shows you how to balance redox reactions under acidic conditions. It also shows you how to identify which half reaction is oxidation and reduction. This video contains plenty of examples and practice problems.
This chemistry video tutorial shows you how to balance redox reactions under acidic conditions. It also shows you how to identify which half reaction is oxidation and reduction. This video contains plenty of examples and practice problems.
Here is a list of topics:
1. Separating a Redox reaction into two half reactions - half reaction method
2. Identifying oxidation and reduction half reactions
3. Balancing the number of oxygen and hydrogen atoms by adding H+ or H2O / water under acidic conditions
4. Balancing the charge by adding electrons to the side with the highest total charge
5. Least Common Multiple Technique to Identify the Number of Electrons to make each half reaction equal
6. Adding the two half reactions to get the net redox reaction
How To balance Redox Equations In Acidic Solution oxidation states | |
| 356 Likes | 356 Dislikes |
| 36,862 views views | 1.16M followers |
| Education | Upload TimePublished on 31 Aug 2016 |
Không có nhận xét nào:
Đăng nhận xét